

Insights and News


The Complexity Spectrum: Configurable and Flexible Approach
The following series of three use cases explores the varying levels of complexity inv...
-1.png)
4G Clinical Appoints Dr. Ronan Brown as Chief Operating Officer
Brown brings years of industry expertise to oversee and maximize 4G Clinical’s operat...
.gif)
Juneteenth
It is an early summer afternoon. The grill is sizzling, sending aromas of meat, sauce...

Somewhere Over the Rainbow-Colored Logo
Once again, the vibrant celebration of Pride Month has arrived! Throughout June, a ka...

Finding Purpose in Clinical Trials: A Personal Reflection on Making a Difference
On this year’s Clinical Trials Day, I pause to reflect on what working in Clinical Tr...
Employee Spotlight: Nicolas Biber
4G Clinical's Employee Spotlight series focuses on showcasing the diverse and talente...
.png)
Inspiring Innovation: Leading Creatively
As a Principal Support Project Lead, my role encompasses more than just overseeing ta...

Berlinger and 4G Clinical Pave the Way to Real-Time Oversight in Clinical Trials
WELLESLEY, Mass., Feb. 27, 2024 – 4G Clinical, a global leader in randomization and t...

Safeguarding Success: Outlining the Crucial Role of Experience in Patient Safety and Trial Success
Within the world of clinical trials, the intersection of experience and innovation is...

Clinical Supply Horror Stories
During this late, spooky time of year, we can look back at some real-life scenarios w...
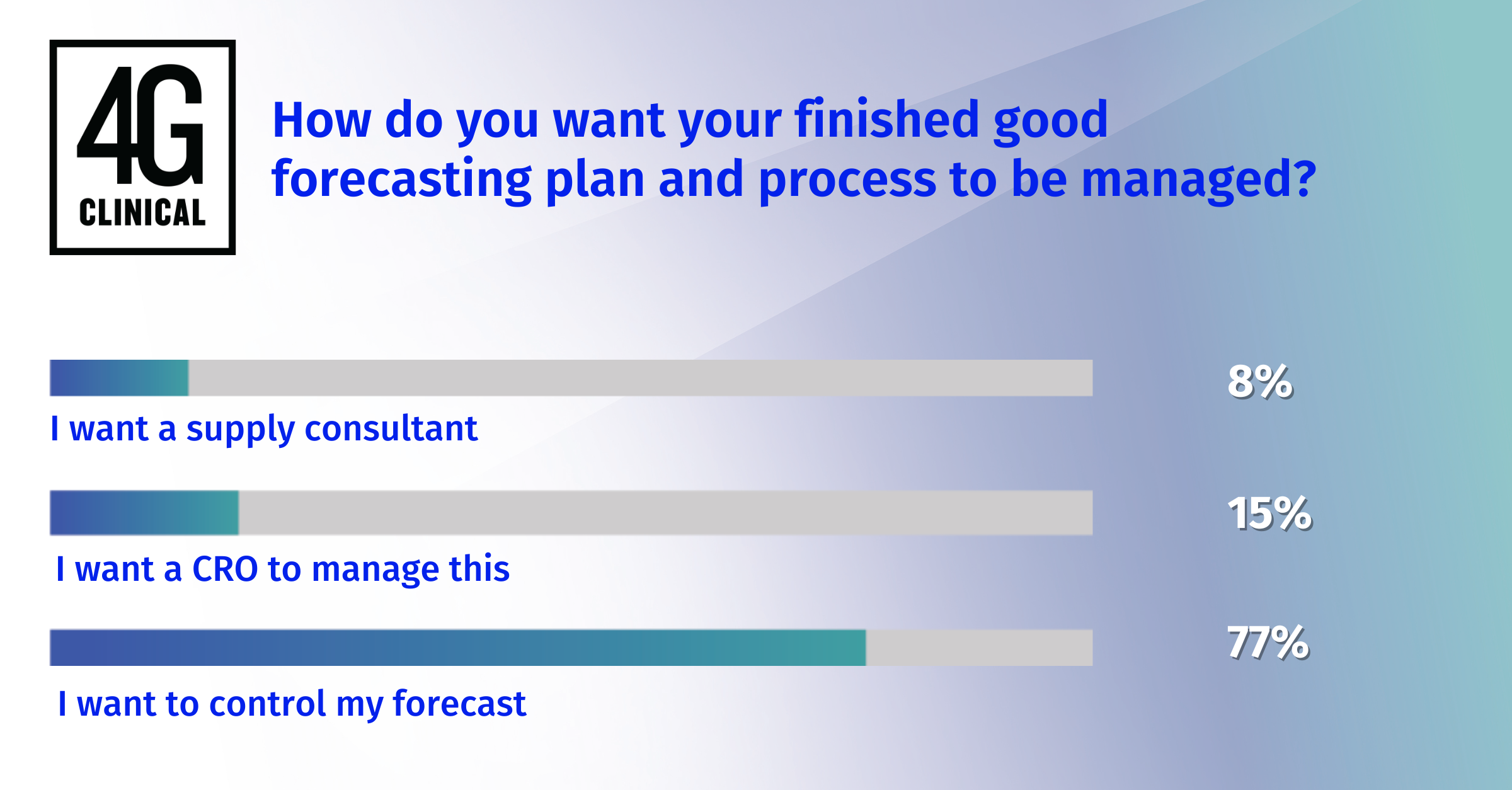
LinkedIn Poll Results: Thoughts From Senior Director of 4C Supply®
4G Clinical recently ran survey polls to clinical supply professionals to collect fee...