The pharmaceutical industry, not unlike many others, is full of acronyms. Some are industry recognized, others are specific to internal organizations. Most of the terms are just different flavors of the same thing. Wouldn’t it be nice if the industry could come together and agree on a uniform term?
We’ve been asked why we use agile RTSM (Randomization and Trial Supply Management) instead of IRT (Interactive Response Technology) when we talk about our software. Before I get into the “why” behind our chosen terminology, I wanted to provide some insight into the evolution of the aforementioned acronyms used throughout the history of these critical systems.
RTSM Through The Ages - 1980s to Now
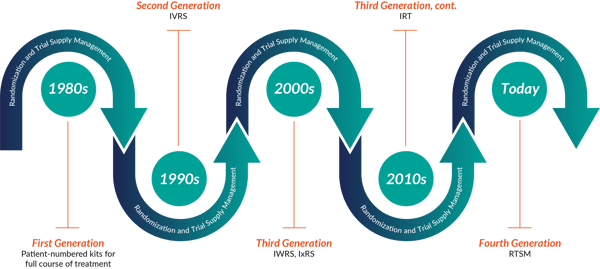
- 1980 - Let’s wind the clock back to the 1980s. Patient-numbered kit labels were shipped to all sites in (generally) full blocks corresponding to the full course of treatments for a study. The sheer quantity of supply wasted in this method by non-enrolling sites, sites that didn’t fill up randomization blocks, and patient dropouts, was staggering.
- 1990 - In the 1990s, the first clinical IVR systems (Interactive Voice Response Systems) were developed in order to randomize patients over the phone, and later, to dispense drug and resupply sites as well. For the first time, all patient kits could be interchangeable for any other equivalent kit.
- 2000 - In the early 2000s, with the advent of the internet, the first parameter-driven, web-based (Interactive Web Response Systems) were born. Now there were two widely used terms, IVRS and IWRS, to describe these systems depending on their modality (phone vs. web).To add to the complication, if an organization used both IVRS and IWRS, the term IxRS was coined to imply either/or.
- Today - Having said that, I would say that the most widely used term in the industry today for our systems is IRT. IRT (Interactive Response Technologies) is considered more of an umbrella term that encompasses all modalities (voice, web and even mobile).
To me, IRT is a very general term that could almost be applied to any application you use to enter data and gather information from, including websites, smart phones, etc. Using the term “IRT” is just like saying “system.”
Why 4G Goes By Agile RTSM Instead of IRT?
So, back to the original question at hand. Why would we choose to go with RTSM to describe our offering and add another acronym to the mix?
Well, as much as we would like to take credit for it, 4G did not coin the term RTSM. RTSM has arguably been around since the advent of these technologies, but it refers more to the function of the actual system versus the modality of delivery.
Let’s look at this this way, historically IRT was used primarily for randomization and getting drug to the site. Over the years, it became apparent that supply management was just as critical as randomization and the combined action of them together elevates the function of this system. They are synergistic and need to co-exist. It also helps to bridge the silos between clinical and supply organizations.
There is a treasure-trove of data housed in RTSM systems that can be used to streamline and accelerate clinical trials – both operationally and to enable supply chain optimization. As trials become more complex, it is more important than ever for clinical and supply to be connected.
For more on this topic, view our blog, Three Generations of RTSM.
Fundamentally, we feel the term RTSM more accurately reflects the core function of the system – and this way, we don’t have to invent a new term for every different modality of delivery that comes next – of which we are sure there will be several.
Interested in discussing this further? Contact Us