Managing protocol versions at the site level and the broader protocol amendment at the sponsor level go hand-in-hand. Neither is simple, but both sides of the equation must be taken into consideration when designing the study.
Let’s take a Master Protocol Trial as an example. The design of the study allows for changes to either the disease sub-types included or to the treatments explored. By keeping the options open, sponsors can follow any positive signals and refine the treatment as data is collected over the course of the study.
The challenge? These changes are usually rolled out via protocol amendments. So not only does the sponsor need to handle challenges resulting from the amendments including supply chains, quality, and timelines, but they also need to have a process for individually applying those amendments to sites that are currently enrolling patients.
Traditionally, sponsors would request their RTSM vendor to create a setting within the RTSM system applying the protocol version change, that the vendor would set manually on a site-by-site basis. This setting is custom-coded within the RTSM as part of the amendment process, adding time, cost, and risk to the process. Time and cost come with the development of the flag itself. Risk enters the equation because it is up to sponsors to make the request and it is a manual implementation process, while operationally, sites cannot implement protocol amendments at exactly the same time. Each site has to perform its own approval process in order to begin working under the new protocol version.
This often results in multiple protocol versions being used at the same time, so it is critical to know what protocol version each site is operating under.
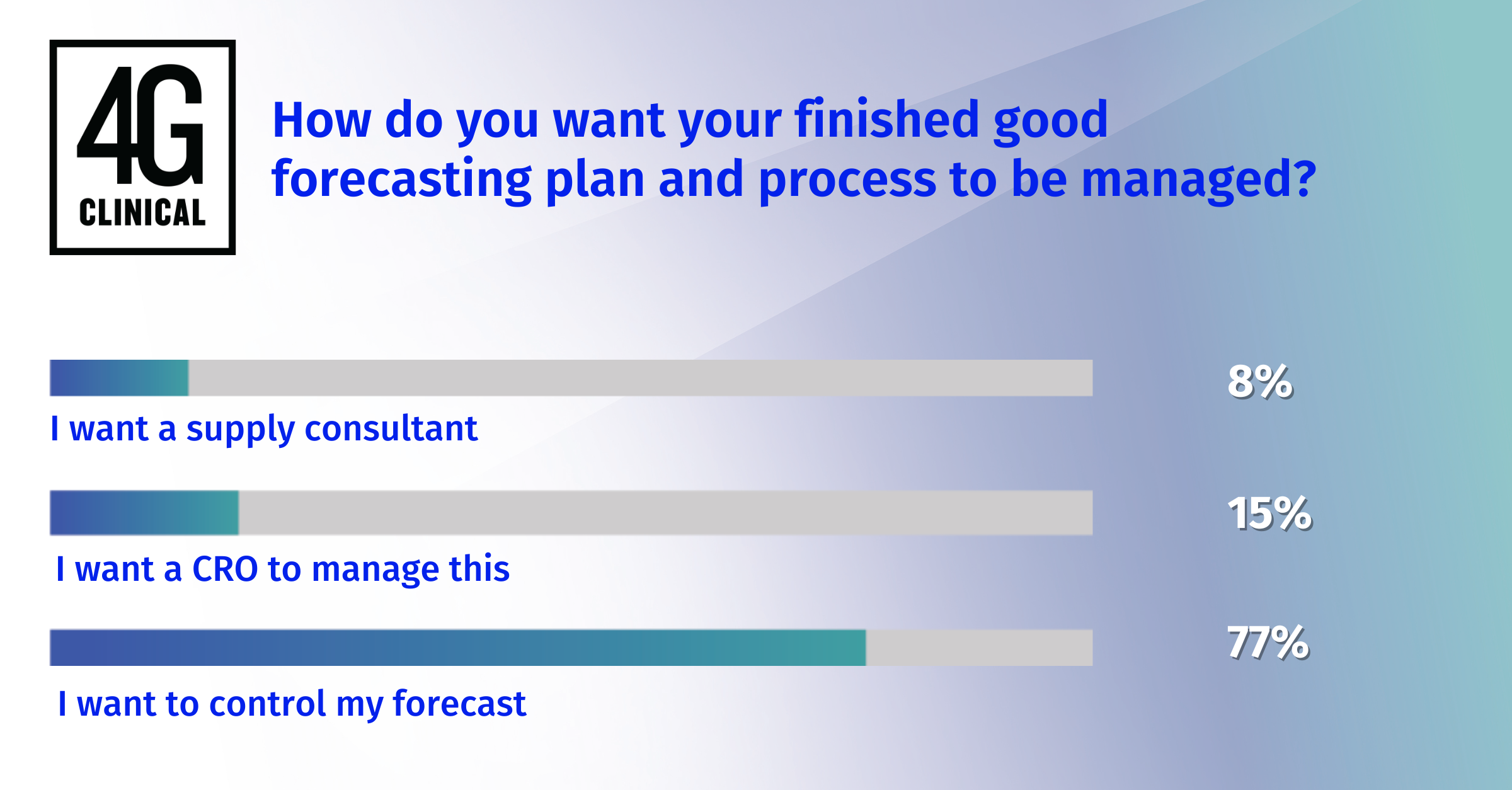
So how can this be managed? Here are three simple steps to manage multiple protocol versions in a single study:
1. Define Protocol Versions Per SiteWhat if you (the trial sponsor) can assign the correct protocol version to the site? Each protocol version can be connected to a different visit schedule, different dosing schedule, specific cohorts, etc., depending on the requirements of the protocol change. This enables any new patient registered and/or any upcoming patient visits to fall under the new protocol version.
2. Simplify the Amendment ProcessAs mentioned above, traditional RTSM vendors custom-code protocol versioning within the system and manually apply to the sites. This process can be error-prone, lengthy, and costly. What if the ability to transition to different protocol versions within a live study was built into the RTSM as a simple configuration? The cost of the amendment decreases, as does the time to implement the change. Additionally, quality is higher as there is less risk of error.
3. Leverage Modern RTSM Systems
Traditional RTSM systems were not built to support the level of flexibility and agility needed from a technology standpoint. Studies with multiple protocol versions are critically dependent on configurable and flexible randomization and trial supply management (RTSM) systems. Not only can you build in flexibility from the onset, but mid-study changes and amendments can become faster and more efficient while offering robust quality.
To learn more, download the Master Protocol webinar recording.